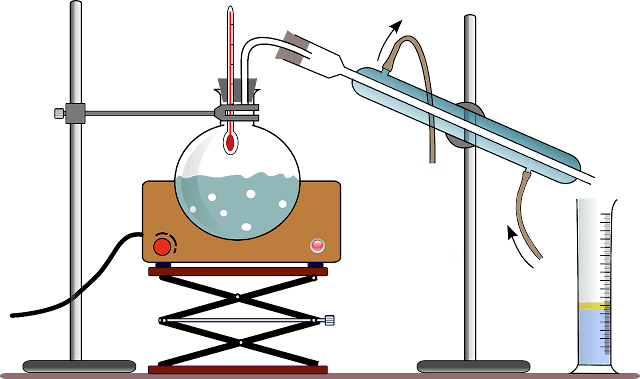
The boiling point of
water increases when the external pressure is increased whereas decreased
external pressure decreases the boiling point. This principle is used in the
process of freeze-drying. Many compounds cannot be distilled at atmospheric
pressure because their boiling points are so high. At their normal boiling
points, the compounds decompose. Thus, some of these materials can be distilled
under reduced pressure because the required temperature to boil the liquid can
be lowered significantly. If the boiling point is lowered by 10 °C each time
the external pressure is halved. To vaporize a liquid, its temperature can be
raised or its pressure can be decreased. Distillation under reduced pressure
can also be called vacuum distillation. During vacuum distillation, the
pressure inside the distillation column is maintained at a vacuum to lower the
temperature needed to vaporize the liquid. This method of distillation is used
for heat-sensitive products, liquids with low viscosities, and liquids that
tend to foul or foam. In vacuum distillation, vacuum pumps are added to the
distillation system to decrease the column pressure below atmospheric pressure,
Fig.1. Careful pressure control is important because the separation is
dependent on the differences in relative volatility at a given temperature and
pressure. Changes in relative volatilities could adversely affect the
separation.
Fig.1: Distillation under reduced pressure
Principle of Distillation Under Reduced Pressure
This distillation method
works on the principle that boiling occurs when the vapor pressure of a liquid
exceeds the ambient pressure. Vacuum distillation is used with or without
heating the mixture.
Construction of Distillation Under Reduced Pressure
The vacuum distillation
unit consists of a distillation column, condensing distillate, and reboiler.
Vacuum pumps and vacuum regulators are added to distillation columns to
maintain the column at a vacuum. Many mixtures can be distilled at much more
economical temperatures with the use of these vacuum distillation columns.
Working of Distillation Under Reduced Pressure
Vacuum distillation is
also called low-temperature distillation. The target product in this
distillation could either be the remaining product, the distilled product, or a
purified product. The vacuum pressure associated with a distillation depends on
the product to be distilled. For example, volatile substances like those used
in oil refineries are likely to undergo vacuum distillations at above 1 Torr, perhaps
20-50 mmHg.
Applications of Distillation Under Reduced Pressure
- The products of normal distillation are further distilled using vacuum distillation. The high boiling point hydrocarbons, such as lubricants and waxes, are separated at economical temperatures.
- Vacuum distillation is also used in the separation of sensitive organic chemicals and recovery of organic solvents.
Advantages of Distillation Under Reduced Pressure
- Vacuum distillation reduces the number of stages needed in distillation.
- The product output per day is very high.
- It increases the relative volatility of the key components in many applications. Lower pressures increase relative volatilities in most systems.
- It requires lower temperatures at lower pressures.
- Vacuum distillation can improve separation by preventing product degradation because of reduced pressure leading to lower tower bottoms temperatures.
- It has a very high capacity to handle liquid mixtures giving a high yield and highest purity.
- It requires low capital cost, at the expense of slightly more operating cost. Using vacuum distillation one can reduce the height and diameter, and thus the capital cost of a distillation column.
- Columns can be operated at lower temperatures.
Disadvantages of Distillation Under Reduced Pressure
- High energy costs of vacuum pumps.
- Pressure and energy losses due to any leaks or cracks.
- Large column diameters are needed for the process to be efficient.
Make sure you also check our other amazing Article on : Flash Distillation - How it Works?