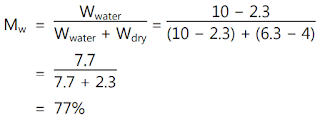The moisture contained in
a material comprises all those substances which vaporize on heating and lead to
a weight loss of the sample. The weight is determined by a balance and
interpreted as the moisture content. As per this definition, moisture content
includes not only water but also other mass losses such as evaporating organic
solvents, alcohols, greases, oils, aromatic components, as well as
decomposition and combustion products. The moisture content is also called moisture
assays which is one of the most important analyses performed on most pharmaceutical
products. Water activity measurements parallel to the moisture content are also
an important parameter for the quality and stability of pharmaceuticals.
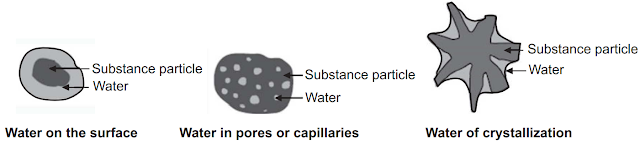
The moisture in products
can be present in different forms based upon the type of bonding with solids,
Fig.1. It is called ‘Free water’ when water is on the surface of the test
substance and it retains its physical form, ‘Absorbed water’ when water is
present in large pores, cavities, or capillaries of the test substance, and
‘Water of hydration’ occluded in lattice ions or water of crystallization
coordinately bonded to ions.
The loss on drying (LOD)
is the amount of water and volatile matters present in a sample when the sample
is dried under specified conditions. Moisture content (MC) is the quantity of
water contained in a material, such as raw materials, API, and blend.
| equation (1) |
| equation (2) |
Fig.1: Types of Bonding of Moisture in Pharmaceutical Products
The moisture content of
solid more than the equilibrium moisture content is referred to as free
moisture (water). It must be noted that during drying only free moisture is
evaporated. The free moisture content (FMC) of a solid depends upon the vapor
concentration in the air solid surface. The moisture contents of solid when it
is in equilibrium with given partial pressure of vapor in the gas phase is
called equilibrium moisture content (EMC). Similarly, the moisture content at
which the constant rate drying period ends and the falling rate drying period
starts is called critical moisture content (CMC). During the constant rate
drying period, the moisture evaporated per unit time per unit area of drying
surface remains constant and in the falling rate drying period the amount of
moisture evaporated per unit time per unit area of drying surface continuously
decreases.
When the water vapor
pressure of the air approaches the saturation water vapor pressure at the
temperature of the gas, the EMC of materials rapidly increases. At these
stages, the process undergone by the material is not only adsorption. Water
vapor begins to condense within the pore structures of the materials.
Theoretically, if the material is in contact with air that is 100 % saturated
for a very long period, all pores of the material should be filled with condensed
moisture. The EMC that corresponds to that hypothetical state is called the
saturation moisture content (SMC) of the material. But in practice, the rate of
this process becomes infinitesimally small at an EMC that is known as the
capillary saturation moisture content (CSMC) and is often substantially less
than the saturation moisture content referred to above.
Measurements
The moisture content is
determined by several direct and indirect methods.
(i) Direct Methods:
The direct methods include mainly thermogravimetric methods. The moisture
content can be determined by an oven method directly. The solid is weighed and
dried, then weighed again according to standardized procedures. In the
Thermogravimetric method, moisture is always separated. Thus, there is no
distinction made between water and other readily volatile product components. A
representative sample must be obtained to provide a useful moisture content
evaluation. Also, the moisture content of the product must be maintained from
the time the sample is obtained until the determination is made by storing it in
a sealed container. Thermogravimetric techniques can be used to continuously
measure the mass of a sample as it is heated at a controlled rate. The
temperature at which water evaporates depends on its molecular environment. The
free water normally evaporates at a lower temperature than bound water. Thus by
measuring the change in the mass of a sample as it loses water during heating
it is often possible to obtain an indication of the amounts of water present in
different molecular environments. For many solids, this method is mandatory,
particularly for granules. For granules, the moisture content is measured by
heating them in a hot air oven at a suitable temperature until the weight
becomes constant. For heat-sensitive materials vacuum is applied in the oven to
decrease the boiling point of the liquid.
(ii) Indirect Methods: Indirect
methods are developed to determine the moisture content rapidly. For example, the
use of modern heating measurement methods like infrared, microwaves, ultrasound,
and spectroscopy. These methods are developed due to requirements of rapid,
non-destructive, and precise moisture content determination. The indirect
methods are generally faster than the direct methods for moisture
determination. When done properly, the indirect methods can be accurate and
precise. However, the accuracy and precision of the indirect methods depend on
careful preparation and analysis of known standards to establish reliable
calibration curves. Indirect methods require a large capital investment in
equipment. Nevertheless, preparation of the standards and accurate calibration
curves must be verified by a specific direct method to establish a reliable
indirect method of instrumentation that can achieve accurate and precise
predicted values.
The methods for moisture
determination given in USP24 NF19 are the best, classical, and addresses only
the determination of moisture content. The U.S.P. offers two methods for the
determination of moisture content in solids:
- (a) Titrimetry (Karl Fisher titration).
- (b) Gravimetric (Thermal gravimetric analysis).
Moisture content is used
in a wide range of scientific and technical areas and is expressed as a ratio,
which can range from 0 (completely dry) to the value of the materials' porosity
at saturation. It can be given on a volumetric or mass (gravimetric) basis.
Moisture content is expressed as a percentage of moisture based on the total
weight (wet basis) or dry matter (dry basis). Wet basis moisture content is
generally used. The moisture content is expressed by the following formulae.
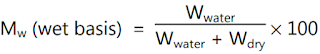 |
| equation (3) |
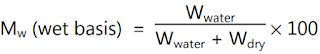 |
| equation (4) |
where,
- M = Moisture content on a percent basis,
- Wwater = Total weight as wet weight
- d = Total as dry weight
Based on the different
forms of moisture present in the material the method used for the measurement
of moisture may estimate more or less moisture content. Therefore, for
different pharmaceutical products, Official Methods of moisture measurement
have been given by agencies.
Example:
Accurately 10 g of granules are transferred into a 4 g container and after
drying the container with granules weighs 6.3 g. What is the percent moisture
content in the granules on a wet basis?
Solution:
Applications of Moisture Content Determination
Accurate percent moisture
content is essential for maintaining the stability of drug products. If a
product is too moist or too dry, it may not be suitable to be administered and
will not exert a desired therapeutic effect. Most pharmaceutical products contain
moisture. The percent moisture content is seldom of interest. Rather, it shows
whether a product intended for trade and production has standard
characteristics such as storage ability, agglomeration, microbiological
stability, flow properties, viscosity, etc. The dry substance content,
concentration or purity, compliance with quality agreements, the therapeutic
value of the product, and legal conformity are other important issues. In
addition, the determination of moisture content has the following applications:
- Freshness:
Fresh products have specified characteristic features. Moisture induces changes
in the state of the solid. As they age and begin to degrade, some dry out and
some pick up excess moisture and begin to mold.
- Labeling:
Pharmaceutical industries require a minimum or maximum percentage of moisture
in certain products for them to be packaged and labeled. If they don't fit these
standards, the products cannot pass the quality standards and are unfit for
commercial release. For example, freeze-dried products, hard gelatine capsules,
etc.
- Cost:
In processed pharmaceutical products, the percentage of water can determine its
final price. Generally, a product with more water will cost less.
- Processing:
The moisture affects the performance of excipients thus manufacturers and
physicians need to know the moisture content of the product to ensure that it
is processed and packaged in a safe, stable way.
- Quality:
Moisture content determines the way most products are appropriate to administer,
taste, feel and look. It is one of the important ways to measure product
quality.
- Shelf life: The physicochemical stability of bioactive agents alone and in combination with excipients is affected by moisture. Thus, the shelf life of the product depends on its moisture content at the time of packaging and the rate of moisture gain during storage. The stability of products depends upon the percent moisture in finished products.
Make sure you also check our other amazing article on : Drying