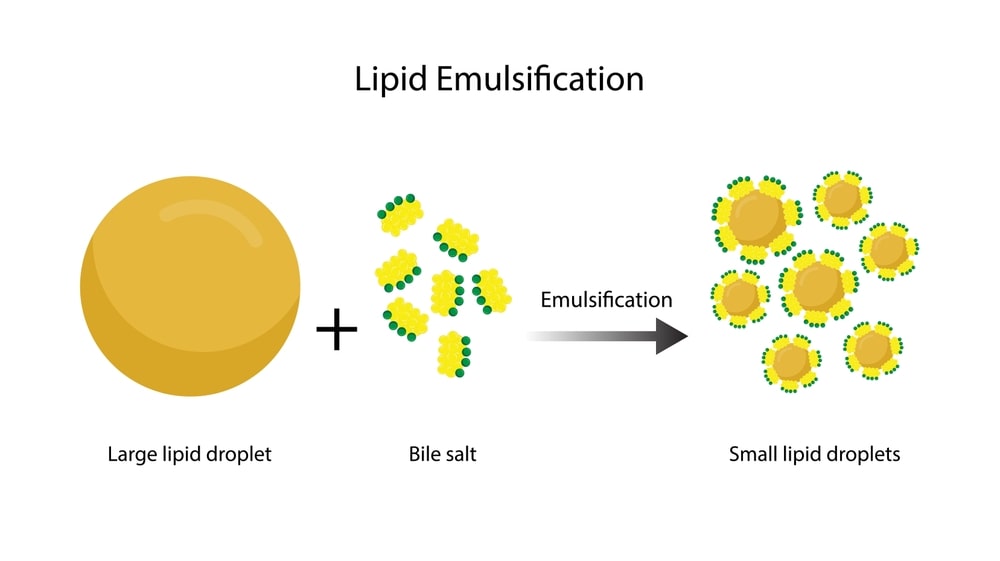
Surfactant:
Adsorbed at oil/water interface to form monomolecular film to reduce the
interfacial tension. e.g., Tween and Spans.
Hydrophilic colloids:
Forming a multimolecular film around the dispersed droplet. e.g., Acacia.
Finely divided solids:
They are adsorbed at the interface between two immiscible liquid phases to form
particulate film. e.g., Bentonite and veegum.
(a) Monomolecular adsorption:
The surface active agent (SAA) is a molecule that has
two parts, one is hydrophilic and the other is hydrophobic. Upon the addition
of SAA, they tend to form monolayer film at the oil/water interface.
The functions of surface active agents to provide
stability to dispersed droplets are as
follows:
• Reduction of the
interfacial tension.
• Form a coherent
monolayer to prevent the coalescence of two droplets when they approach each
other.
• Provide surface charge
which causes repulsion between adjusted particles.
Bancroft rule
As per the Bancroft rule, the emulsifying agent being
used in an emulsion should be favorable to the external phase of the
emulsion.
So even though there may be a formula that is 60% oil
and 40% water, if the emulsifier chosen is more soluble in water, it will
create an oil-in-water system.
The Hydrophilic-Lipophilic Balance (HLB) of a
surfactant can be used to determine whether it is a good choice for the desired
emulsion or not.
In Oil-in-water emulsions, use emulsifying agents that
are more soluble in water than in oil (High HLB surfactants).
In Water in Oil emulsions, use emulsifying agents that
are more soluble in oil than in water (Low HLB surfactants).
(b) Multimolecular adsorption
|
|
Polysaccharides |
Amphoteric |
Synthetic or semi-synthetic polymers |
|
Colloids |
Acacia Agar Alginic acid Carrageenan Guar gum Karraya gum Tragacanth |
Gelatin |
Carbomer resins Cellulose ethers Carboxymethyl
chitin PEN-n (ethylene
oxide polymer) |
Hydrophilic colloids form multimolecular adsorption at the oil/water interface. They have a low effect on the surface tension.
• Their main function as emulsion stabilizers is to
make a coherent multi-molecular film. This film is strong and resists the
coalescence. They have, also, an auxiliary effect by increasing the viscosity
of the dispersion medium.
(c) Solid particle adsorption
Finely divided solid particles are adsorbed at the
surface of the emulsion droplet to stabilize them. Those particles are wetted
by both oil and water (but not dissolved) and the concentration of these
particles forms a particulate film that prevents the coalescence.
|
Finely divided
solids |
Bentonite Hectorite Kaolin Magnesium
aluminium silicate Montmorillonite Aluminium
hydroxide Magnesium
hydroxide Silica
|
Emulsion Stability
The process by which an emulsion completely breaks is
generally considered to be governed by
four different droplet loss mechanisms, i.e.
• Brownian flocculation,
• Creaming,
• Sedimentation flocculation and disproportionation.
Fig: Mechanism leading to coalescence of an oil in water emulsion
Creaming – upward and downward
• Creaming derives its name from the most commonly known example of a demulsification process.
• The separation of milk into its cream and skim milk
components. Creaming is not an actual breaking but a separation of the emulsion
into two emulsions, one of which (the
cream) is richer in the disperse phase than the other. Creaming is the
principal process by which the disperse phase separates from an emulsion and is
typically the precursor to coalescence.
• The creaming rate (or settling rate for dispersed
phases more dense than the continuous
phase) can be estimated from Stoke's equation:
υ = 2r2 (ρ − ρ0) g/9η
where υ is the creaming (settling) rate, r is the
droplet radius, ρ is the density of the droplet, ρo is the density of the
dispersion medium, η is the viscosity of the dispersion medium (continuous
phase) and g is the local acceleration due to gravity.
Flocculation
• The aggregation of droplets to give 3-D clusters
without coalescence occurring.
Importantly, all droplets maintain their own integrity and remain as
totally separate entities. It results when there is a weak, net attraction
between droplets and arises through various mechanisms.
• Flocculation may be subdivided for convenience into
two general categories: that resulting from sedimentation aggregation and that
from Brownian motion aggregation of the droplets.
Disproportionation or Ostwald ripening
• It is dependent on the diffusion of dispersed phase
molecules from smaller to larger droplets through the continuous phase.
• The pressure of dispersed material is greater for
smaller droplets than larger droplets as per the Laplace equation.
Coalescence
• A few globules tend to fuse with each other and form
bigger globules.
• In this process, emulsifier film around the globules
is destroyed to some extent.
Breaking
• Complete separation of phases, irreversible
process.
Phase Inversion
In phase inversion o/w type emulsion changes into w/o
type and vice versa. It is a physical
instability.
• It may be brought about by the addition of an electrolyte, by changing the phase volume ratio by temperature changes, or by changing the chemical nature of the emulsifier Phase inversion can be minimized by using the proper emulsifying agent in adequate concentration, and by storing the emulsion in a cool place.